Inside the Control Tower
Our 17-tab dashboard manages every aspect of your FDA journey — from a guided setup wizard through dual-track development, FDA communications automation, and team collaboration.
▶ 510(k) Lifecycle Walkthrough
See how the Control Tower guides a medical device from Pre-Sub through FDA clearance — step by step.
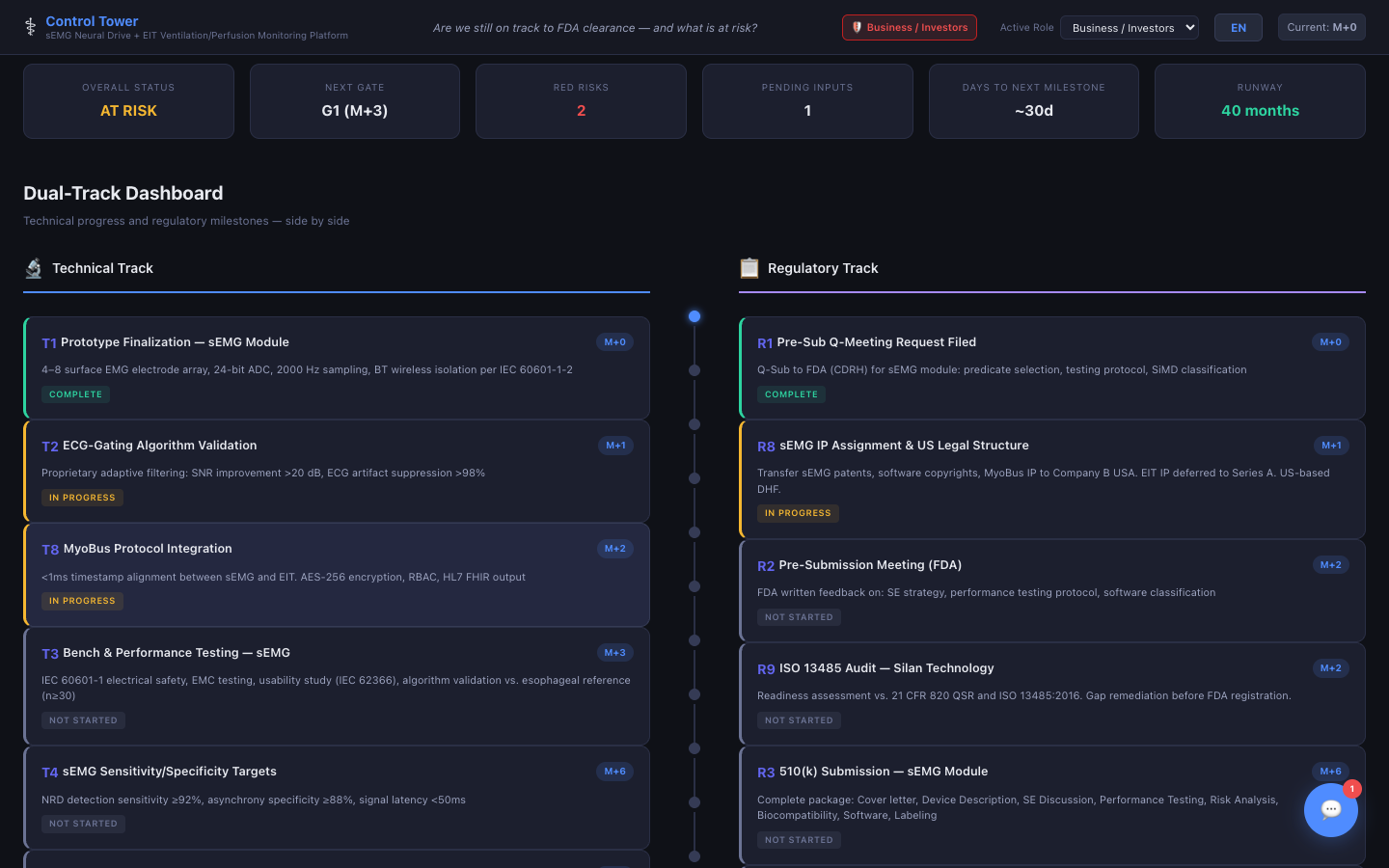
Dual-Track Dashboard
Technical and regulatory milestones tracked side by side — always know where both tracks stand.
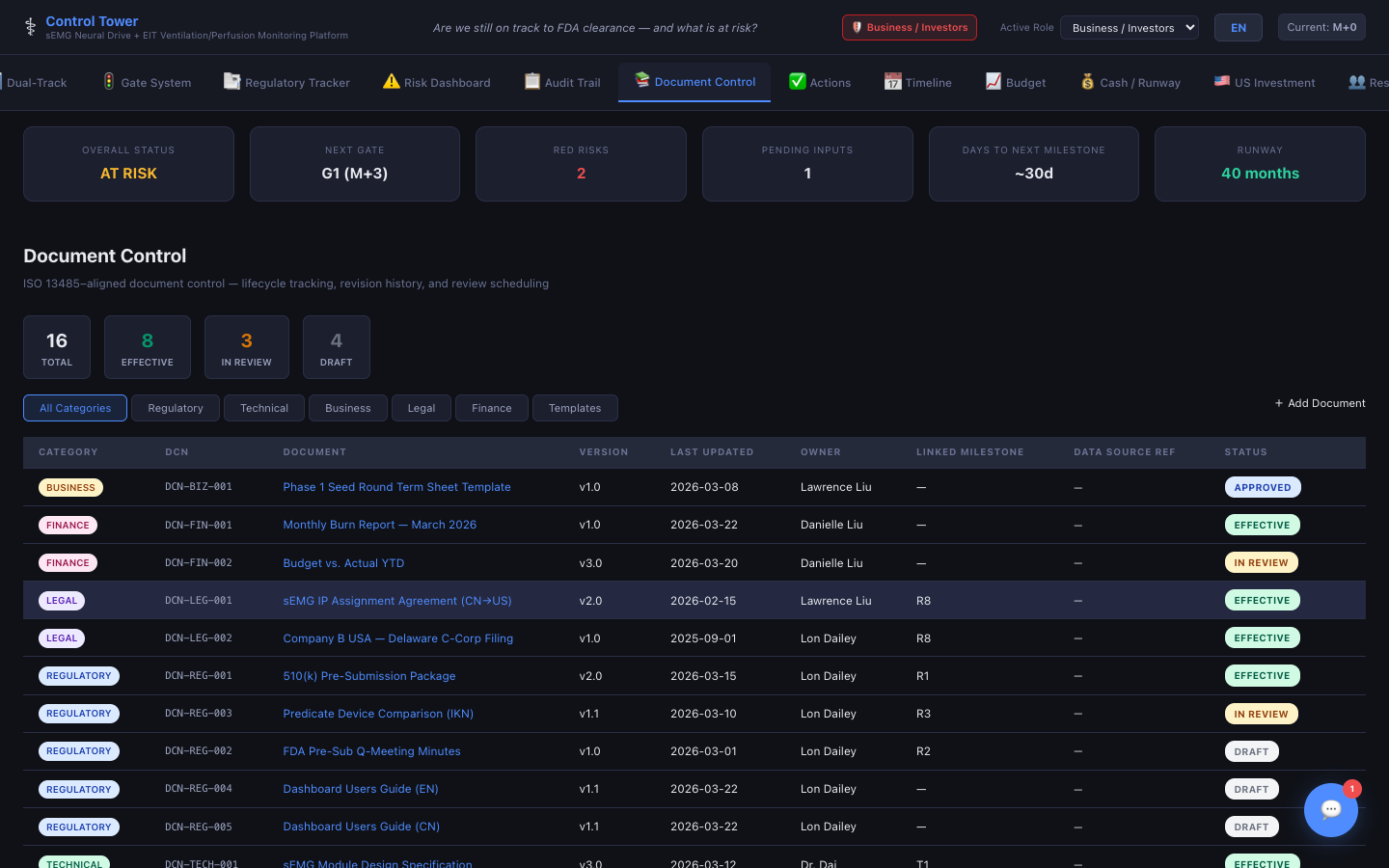
Document Control
ISO 13485-aligned lifecycle tracking with version history, review scheduling, and audit traceability.
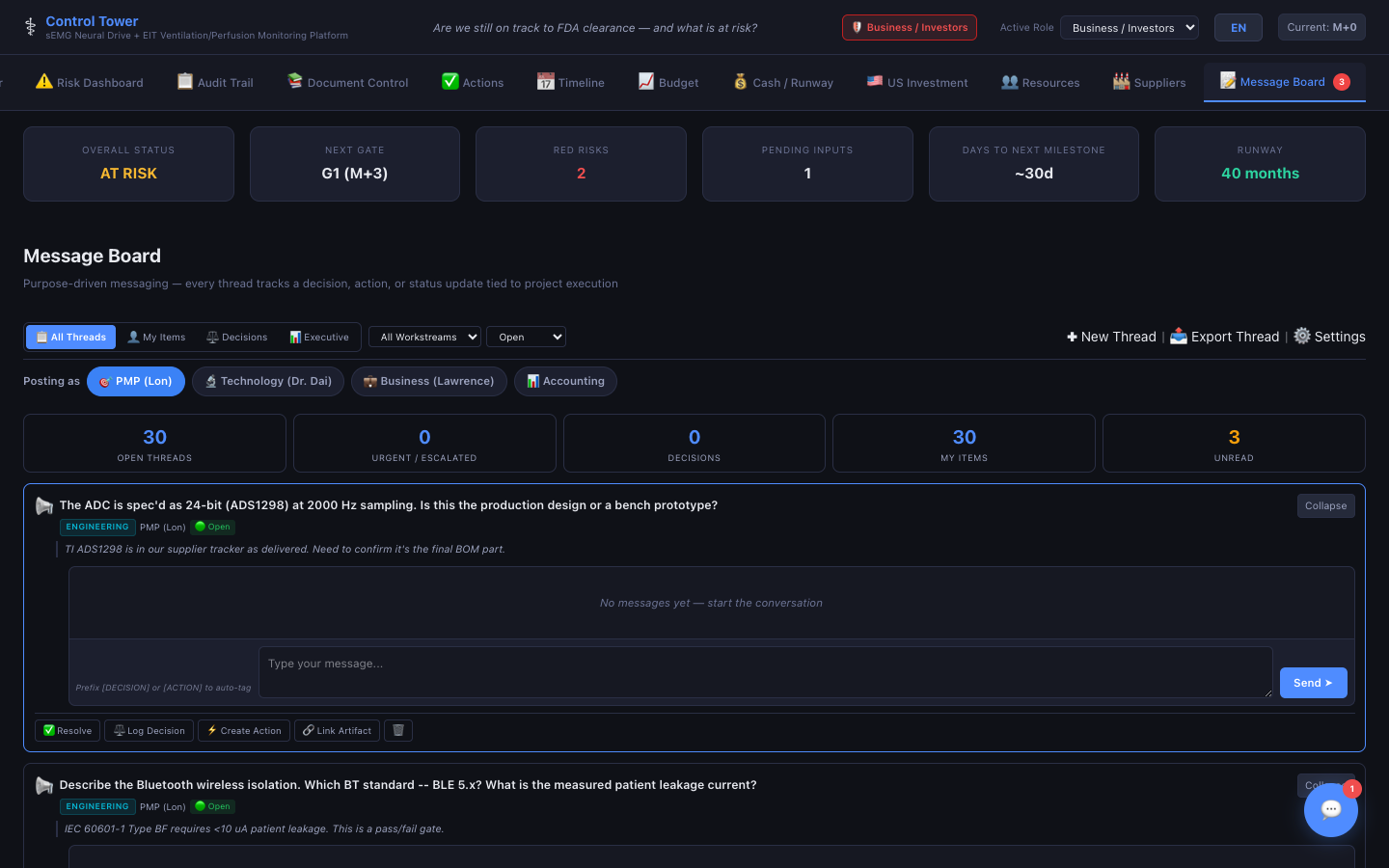
Message Board
Purpose-driven messaging — every thread tracks a decision, action, or status update tied to project execution.
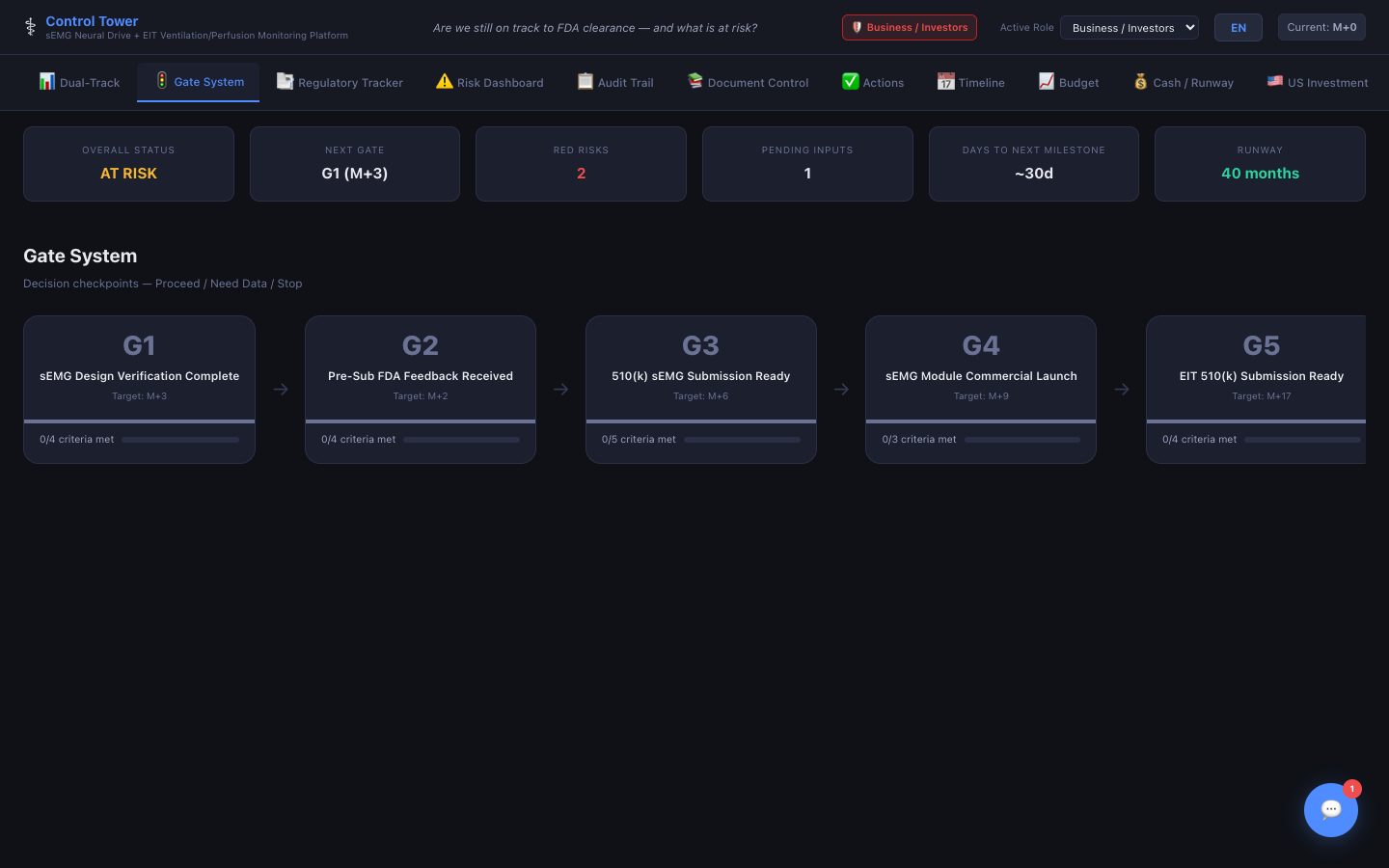
Gate System
Phase-gate reviews with criteria checklists — Go/No-Go decisions recorded with stakeholder inputs and conditions.
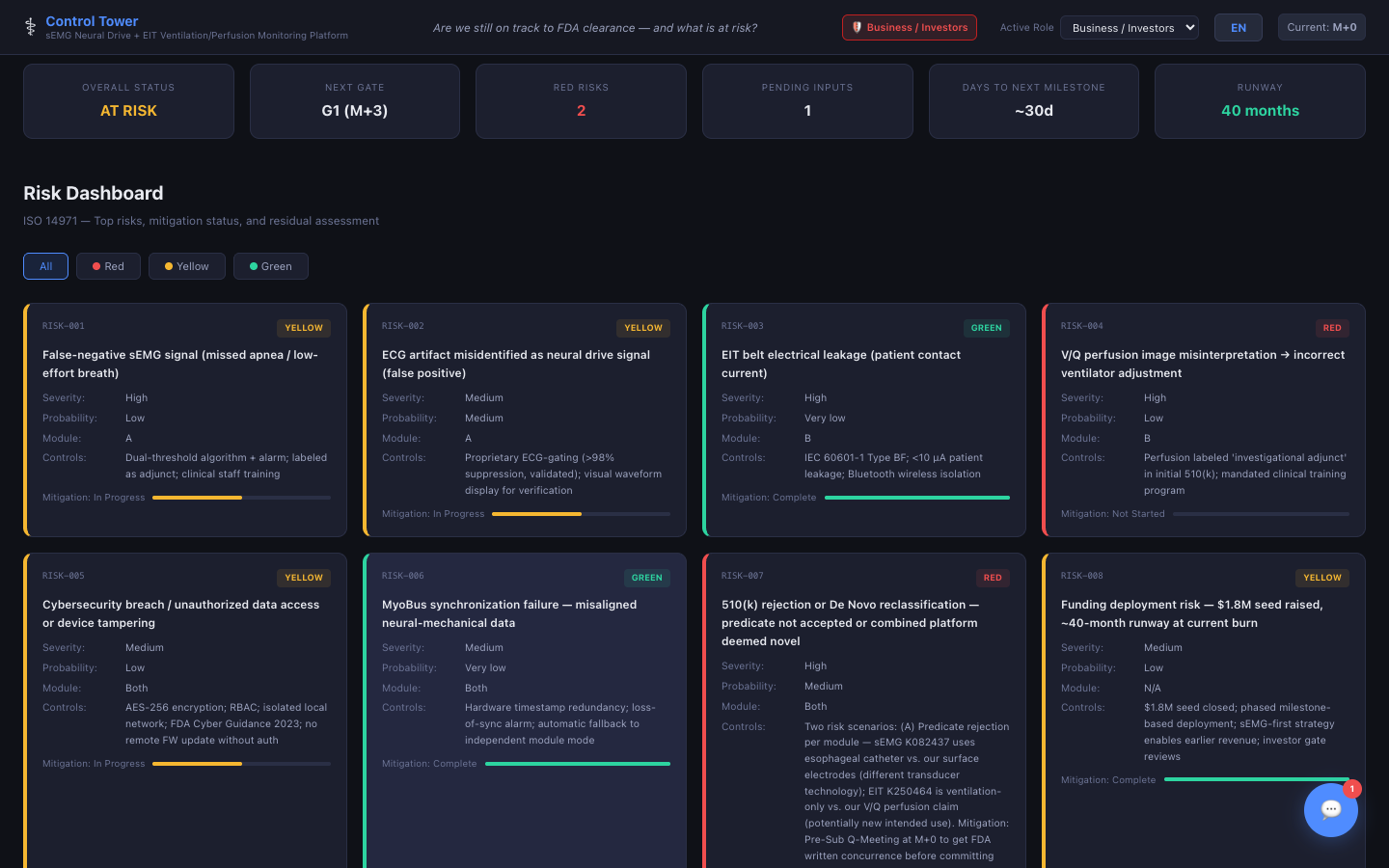
Risk Dashboard
ISO 14971 risk matrix with severity, probability, and color-coded risk levels — track mitigations from identification to closure.
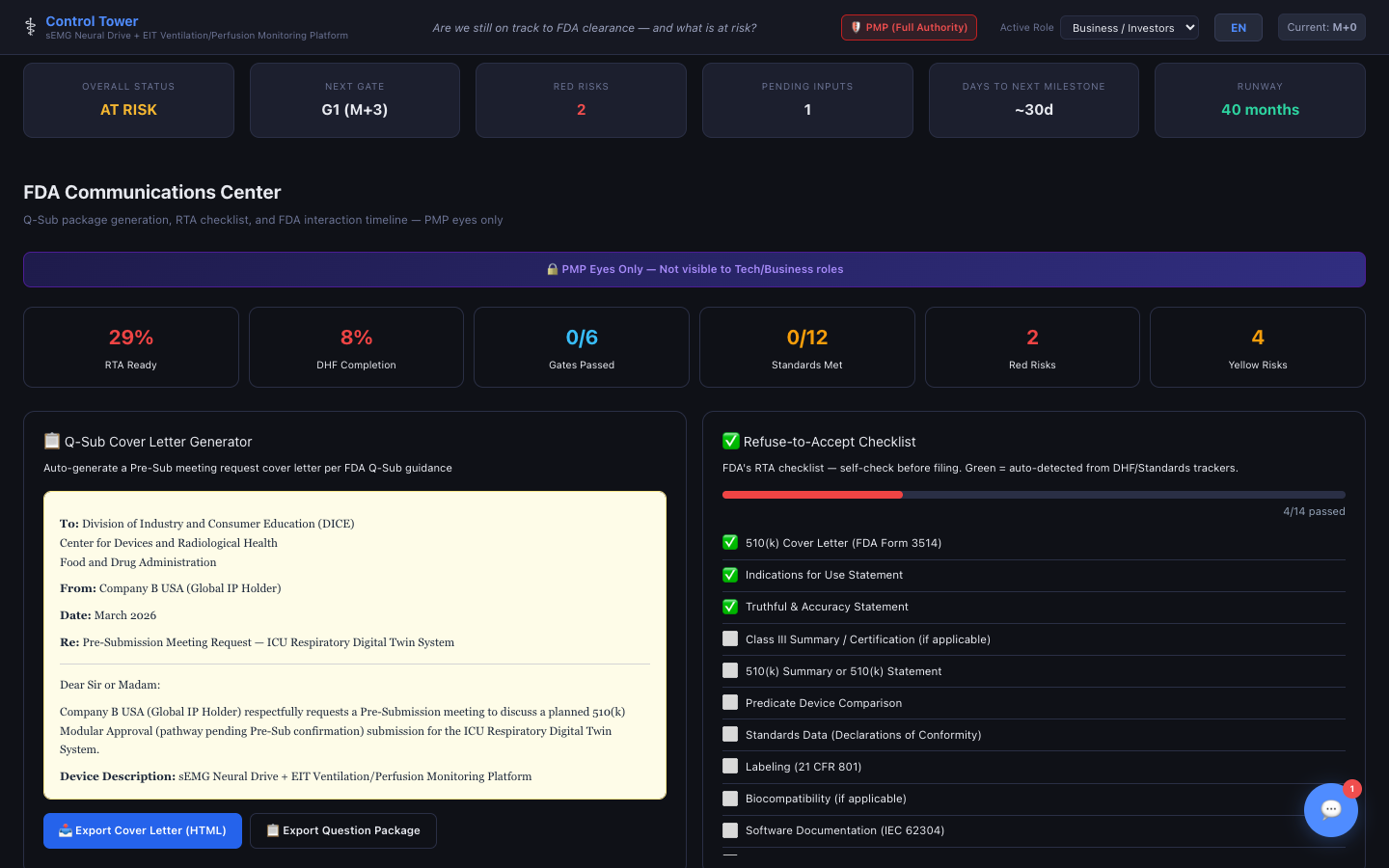
FDA Communications Center
Q-Sub cover letter generator, 17-item RTA self-check, MDUFA 90-day radial clock gauge (color-coded Day 15 RTA → Day 60 review → Day 90 decision, with clock-pause tracking), SE decision flowchart, plus a mini day-badge on the Timeline tab — all in one platform.
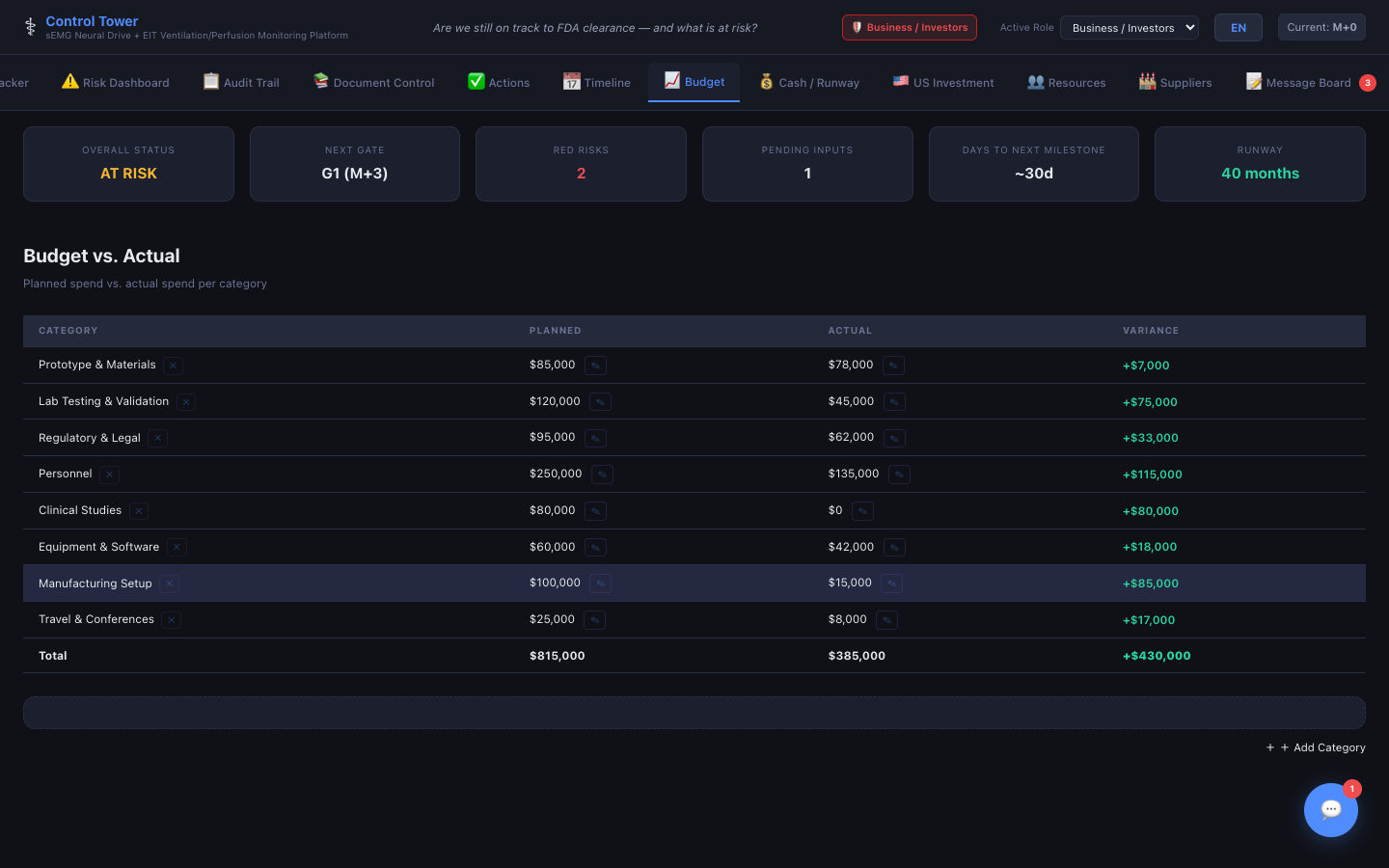
Budget Tracking
Planned vs. actual spend by category with automatic variance calculation — toggle between USD and CNY display.
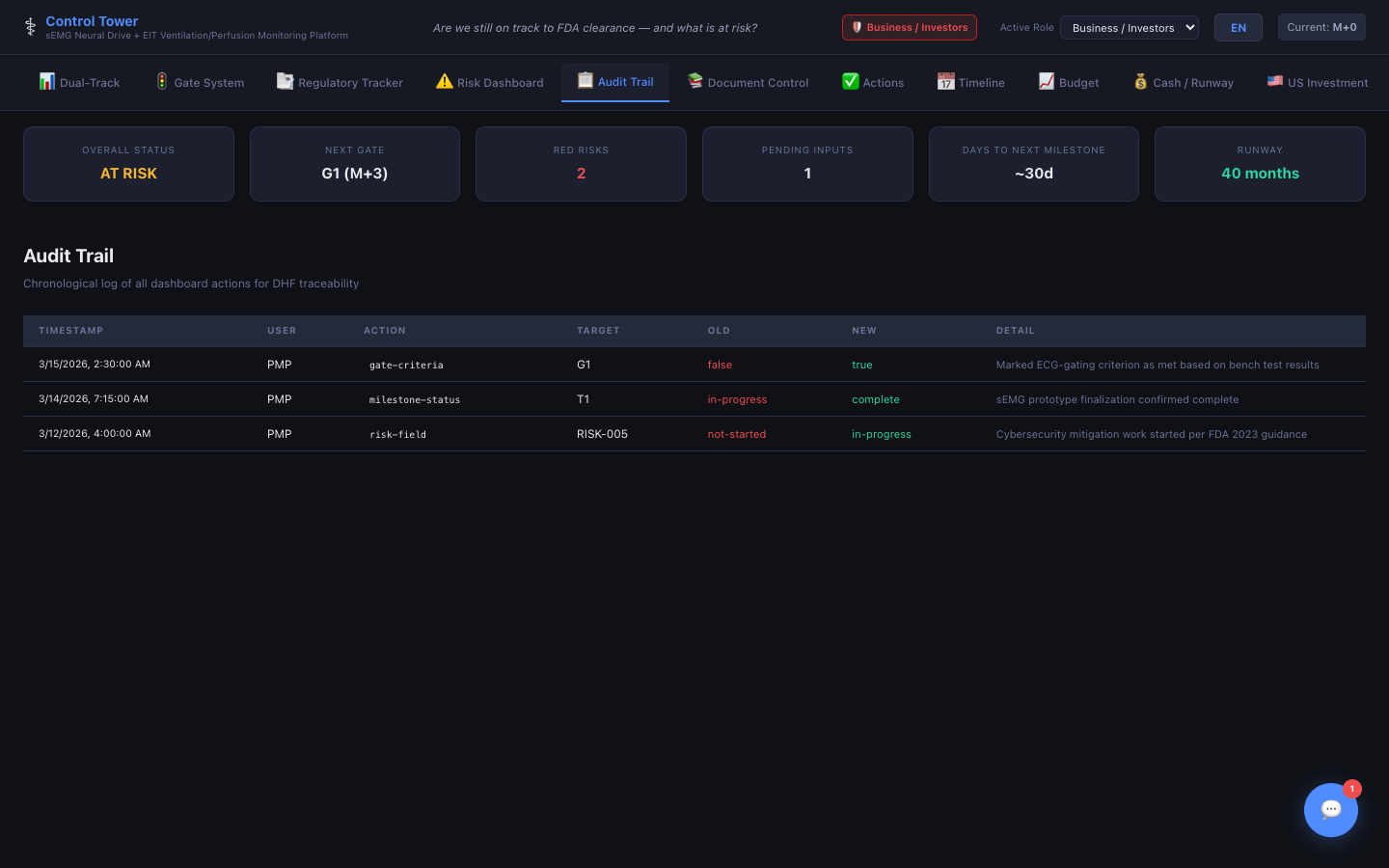
Audit Trail
21 CFR Part 11 compliant — every change timestamped with user, field, old value, new value, and detail description.
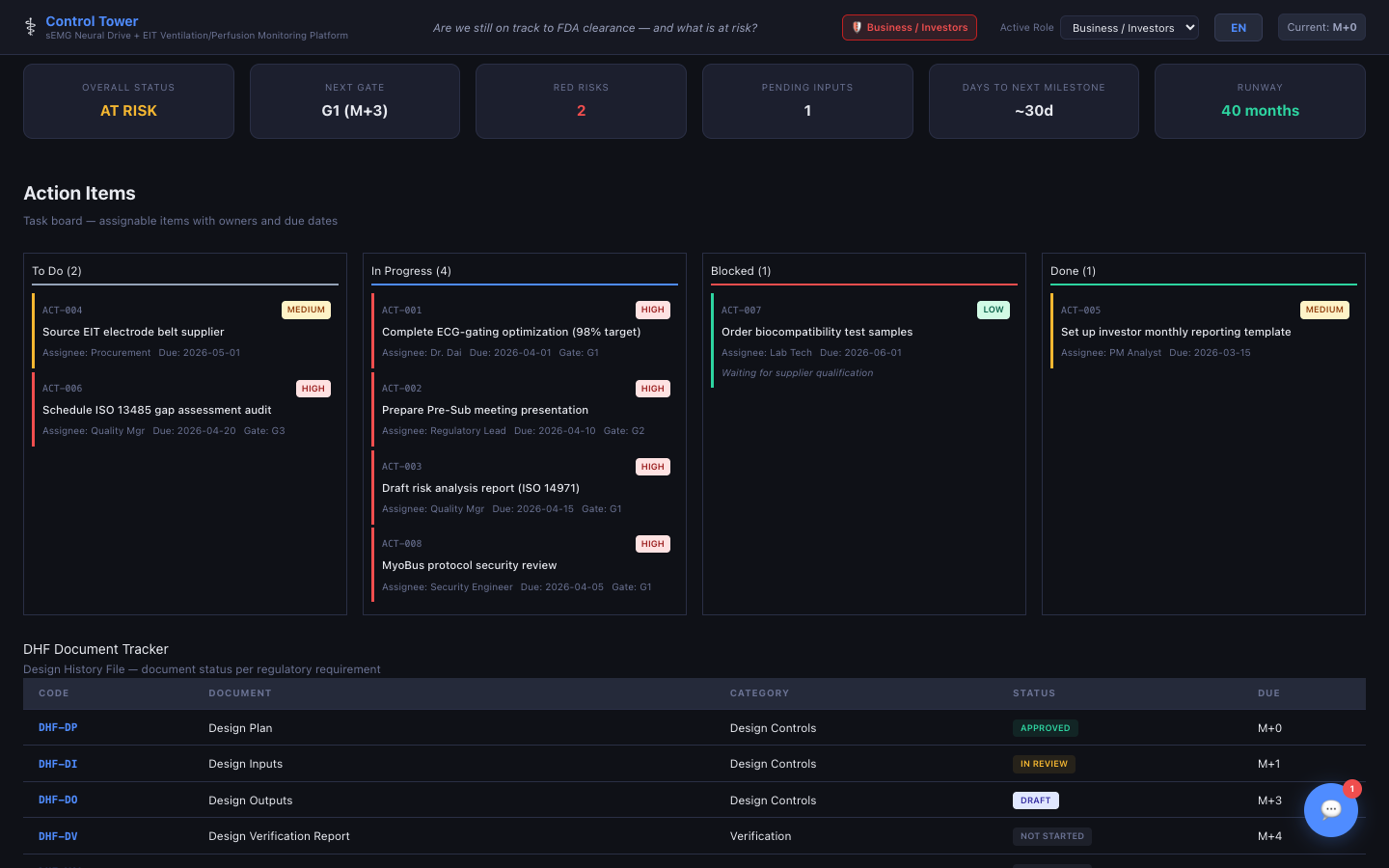
Actions / DHF / CAPA
Task board, Design History File tracker, Device Master Record tracker, and Corrective & Preventive Action log.
All 15 Tabs — Complete Project Visibility
 Dual-Track
Dual-Track
 Gate System
Gate System
 Risk Dashboard
Risk Dashboard
 Audit Trail
Audit Trail
 Document Control
Document Control
 Actions
Actions
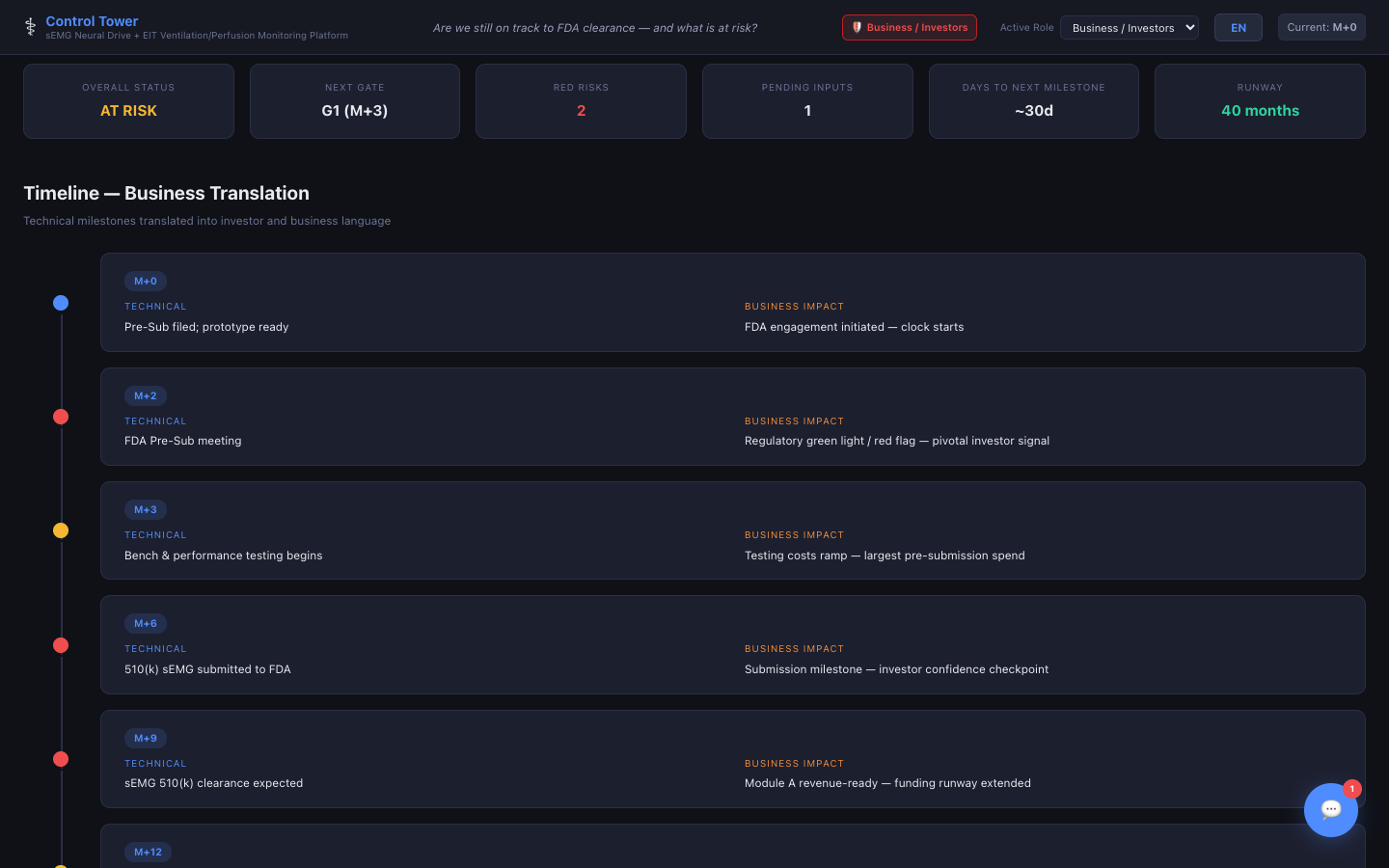 Timeline
Timeline
 Budget
Budget
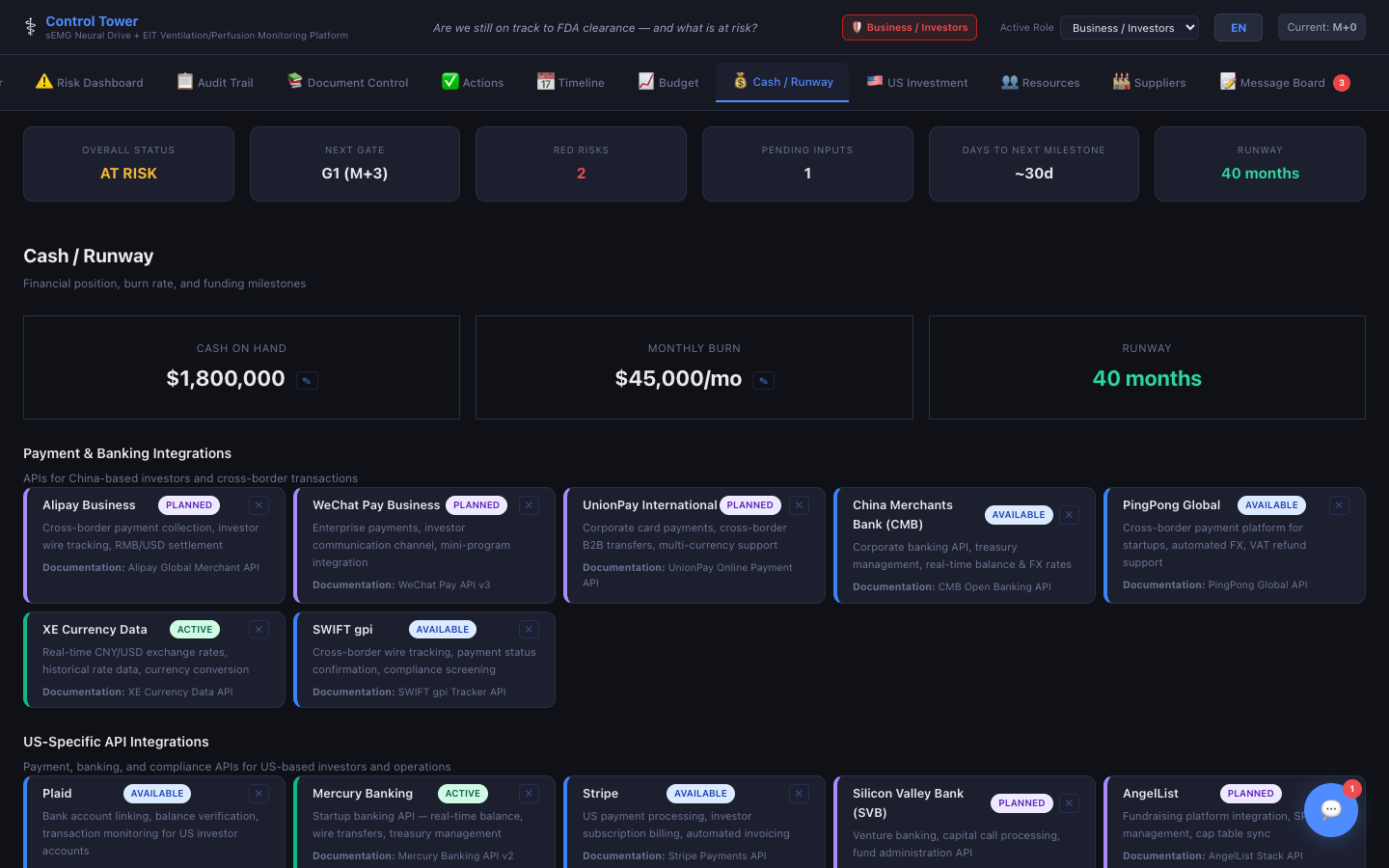 Cash / Runway
Cash / Runway
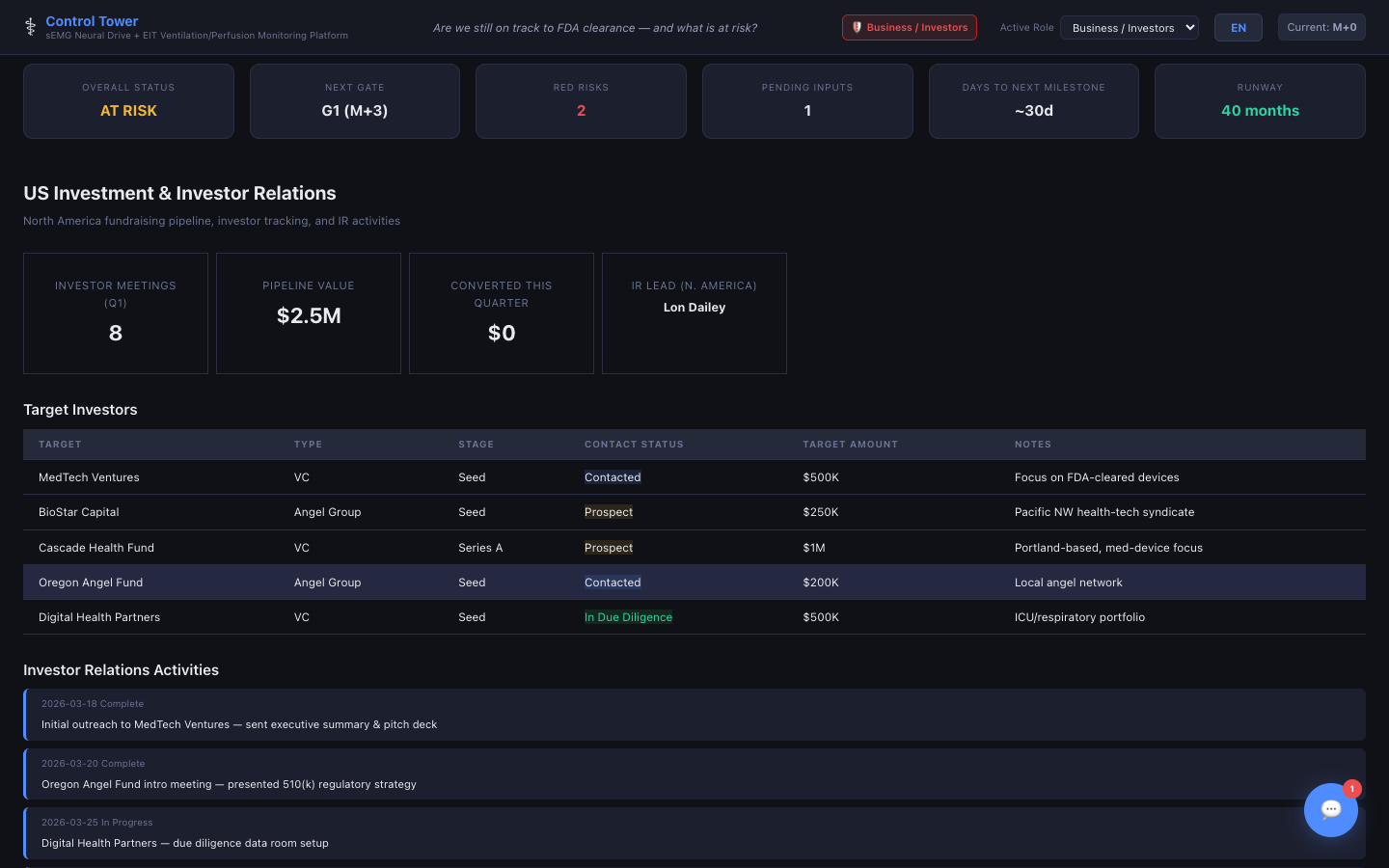 US Investment
US Investment
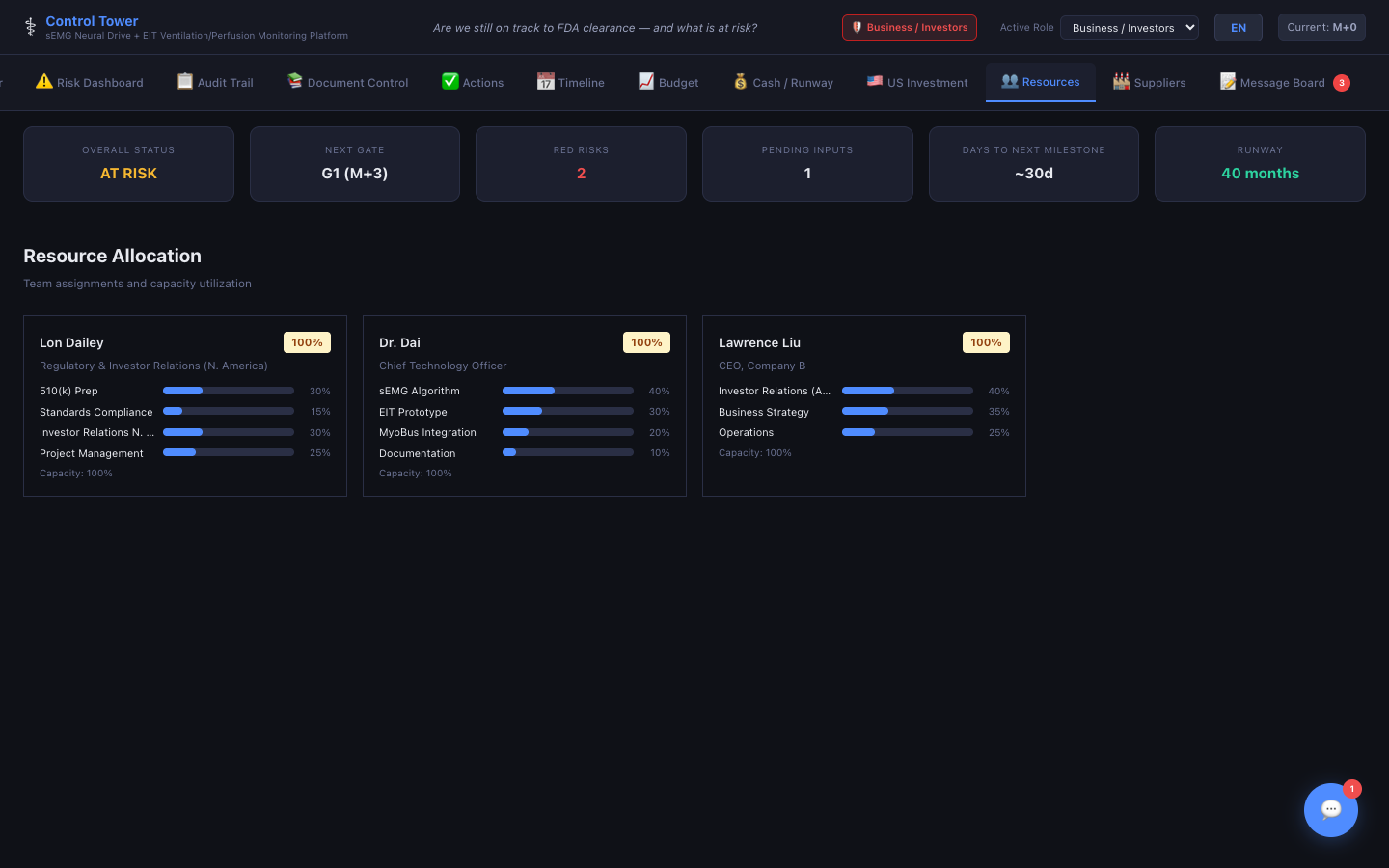 Resources
Resources
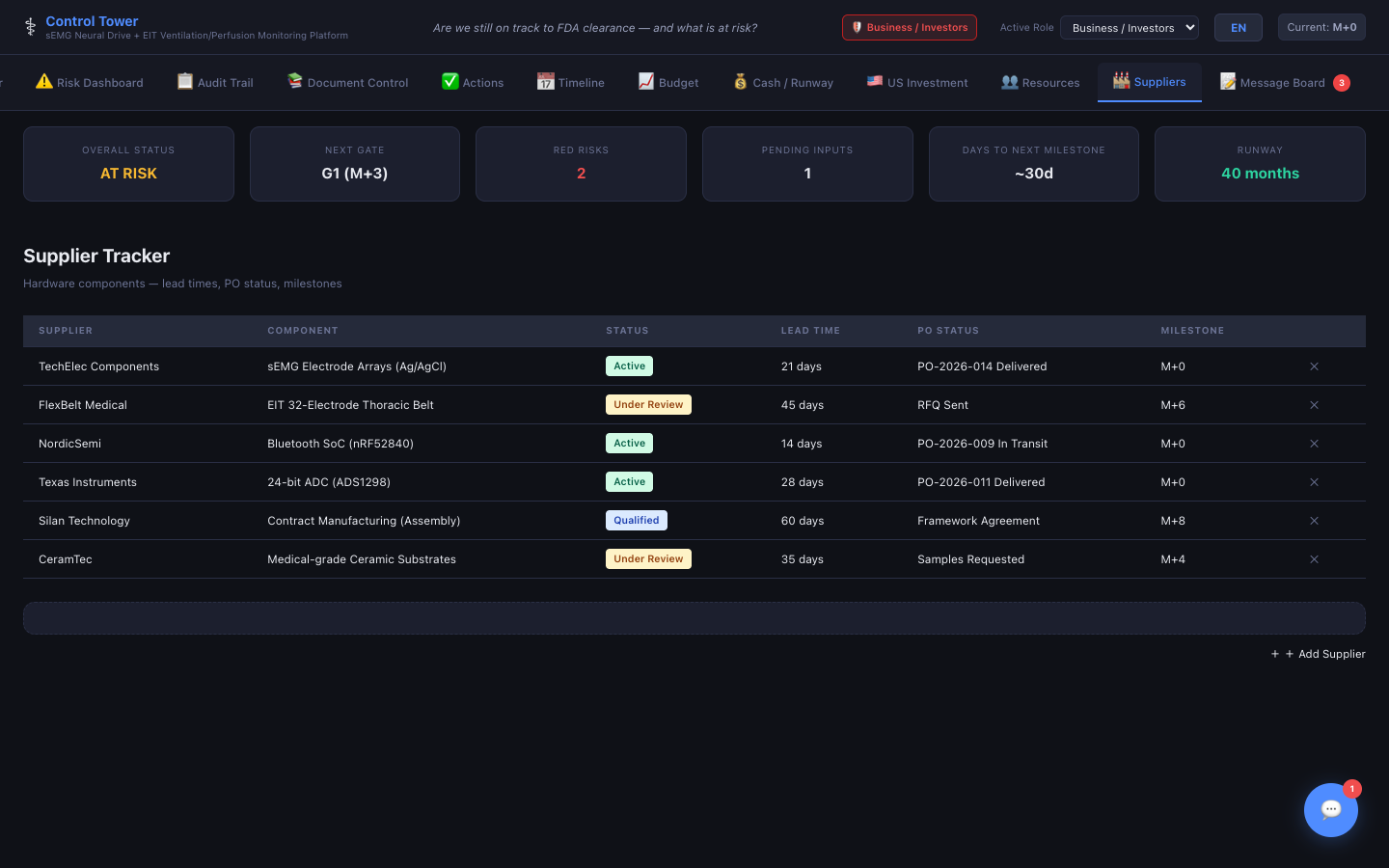 Suppliers
Suppliers
 Message Board
Message Board
 FDA Comms
FDA Comms
